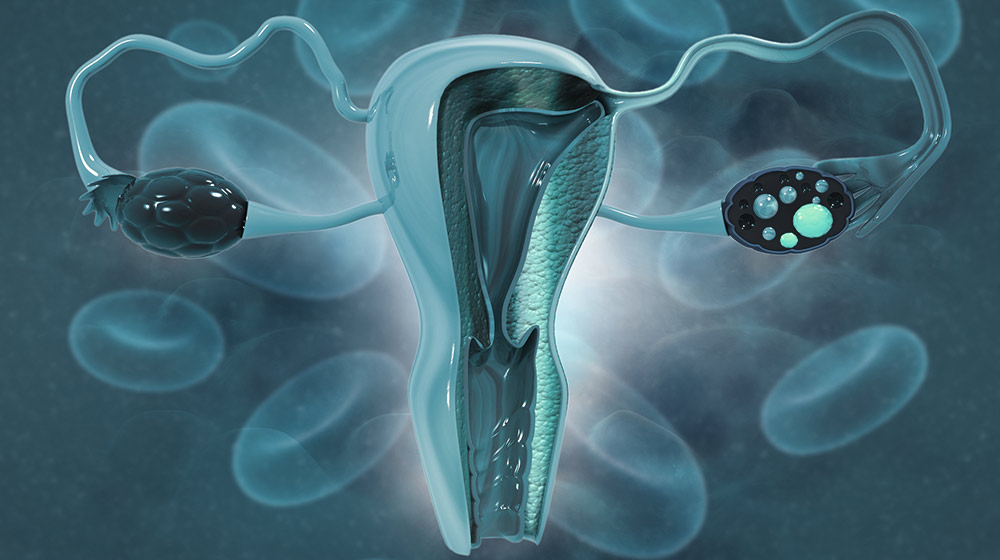
Polycystic ovary syndrome (PCOS), a hormonal disease characterized by irregular menstrual periods, elevated testosterone levels, or enlarged ovaries with many cysts, is a significant cause of infertility in women, affecting up to one in every five women of reproductive age. However, the fundamental processes and causes of PCOS are still unknown.
According to research published this week (February 3) in Cell Metabolism, mice can pass on PCOS-like symptoms for at least three generations. This is most likely passed by epigenetic changes, which are handed from parents to children like a set of instructions directing which genes are to be expressed.
In some genome-wide association studies of PCOS susceptibility, genetics account for less than 10% of the condition’s heredity. According to certain genome-wide association studies of PCOS susceptibility, genetics account for less than 10% of the condition’s heredity. The researchers also examined blood samples from women with PCOS, discovering epigenetic changes comparable to those seen in mice models.
Previously, developmental neuroscientist Paolo Giacobini of the French National Institute of Health and Medical Study and his colleagues discovered a possible etiology of PCOS in prior research. Injecting extra anti-Müllerian hormone (AMH) into pregnant mice caused their offspring to acquire PCOS signs such as increased testosterone, erratic reproductive cycles, and smaller litters.
In the current study, he and his colleagues wanted to see if mice might pass on PCOS-like symptoms to future generations. To do this, scientists married male and female mice whose mothers had been exposed to AMH in the womb, then mated their female offspring with males exposed to AMH in utero, resulting in the third generation of mice.
Read the original publication of this study here: [Polycystic Ovary Syndrome May Be Inherited Epigenetically]
Learn what epigenetics research say about the hereditary factors of polycystic ovary syndrome
Polycystic Ovary Syndrome May Be Inherited Epigenetically
The researchers discovered a smattering of genes with atypical methylation patterns, typically with startlingly low levels of methylation when compared to control mouse methylation profiles. The most impacted gene pathways were those involved in the regulation of reproductive and metabolic processes, such as insulin signaling and inflammation.
Tet1, which encodes ten-eleven translocation methylcytosine dioxygenase, an enzyme necessary for methylation removal, was among the methylation-depleted genes. Researchers also ran DNA tests on blood samples from more than 30 women with PCOS, some of whom were the offspring of PCOS moms.
Interestingly, they discovered that Tet1 was one of the numerous genes that displayed the identical methylation-deprived pattern that they had seen in mice. They have come to a theory that [Tet1] is one of the central genes whose mutation may be responsible for the hypomethylation seen in both PCOS mice and PCOS women. The most exciting discovery is that they can translate this and detect at least some of these epigenetic alterations in women’s blood with PCOS.
The earlier research has also discovered epigenetic alterations in PCOS patients. Such findings might pave the path for the development of possible biomarkers to predict PCOS. To support this idea, the researchers observed that injecting third-generation females with a naturally occurring, methylation-promoting chemical called S-adenosylmethionine might correct some of the symptoms.
Surprisingly, following two or three weeks of therapy, this epigenetic medication was able to restore these animals’ ovulation as well as dramatically ameliorate their metabolic abnormalities.
During the research, it was found that there is a striking similarity between the AMH-induced mouse model and human patients and the fact that the epigenetic changes were so robustly sustained across three generations.
It also supports the concept that AMH plays a direct role in the etiology of PCOS rather than being a result of PCOS. In addition, it serves as a warning about the tremendous impact hormone exposure may have on a multigenerational time frame. For humans, this implies that a woman’s hormone exposure will continue to affect her grandchildren and great-grandchildren for the next 50 years or more.
Takeaways:
- The result of the study conducted on the mice model has answered the question of whether the “Polycystic Ovary Syndrome can Be Inherited Epigenetically,” which is “yes.” Not only that, but it also has raised various research questions about the potential study that could lead to some scientific breakthroughs.
- One unanswered question that researchers aim to answer is whether similar epigenetic changes may also affect male offspring.
- If PCOS is caused by changes in the expression of key epigenetic factors such as Tet1, then all progeny may be affected. So far, this is due to the fact that physicians seldom follow up on the health of boys born to PCOS mothers, much alone track them over time.